Activity 6: Balancing chemical equations
- Due Feb 1, 2016 at 11:59pm
- Points 10
- Questions 10
- Time Limit None
- Allowed Attempts 3
Instructions
110_Activity_Balancing Equations.docx
Background information and purpose:
Chemistry is the study of matter and its transformations. One example of a transformation is when liquid water freezes to make solid water (ice). Liquid, solid and gas are three physical states. When a substance changes from one physical state to another, it's an example of a physical change:
H2O is water. The equation H2O (l) --> H2O (s) means liquid water changes to solid water. This is a physical change since water is present before and after the change.
Physical changes aren't the only changes of matter. Matter can also change from one substance to a completely different one. These are called chemical changes (or chemical reactions).
Chemical reactions are written as equations. For example, for the sentence: Iron rusts by oxidation (reaction with oxygen) to form iron oxide, the chemist would write the equation to describe the process:
Fe (iron) + O2 (oxygen) ---> Fe2O3 (rust)
You will be looking at some of these equations and will apply a fundamental principle of chemistry/nature. If you don't know what that is, don't look it up yet. You will find out by playing with the simulation!
If you have trouble with this assignment, here's a reference:
Read pages 51-53 in Science 101: Chemistry
And here's a video for beginners (3:26): https://www.youtube.com/watch?v=Q4WWiAm3nPM
PRACTICE FIRST! Before starting this activity, please practice.
Here is a practice worksheet (answers are on the second page!): http://www.personal.kent.edu/~cearley/ChemWrld/review/balance.pdf
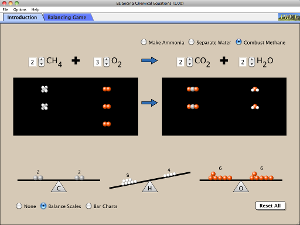
Find the simulation online:
Click the image below or go to the University of Colorado – Boulder PhET website at: http://phet.colorado.edu
Click “Play with Sims”, then click “Chemistry” on the left-hand side menu. Find the “balancing chemical equations” Simulation and click RUN (or you can download it if you prefer).This simulation requires Java – if you don’t have it, install it for free at http://java.com/en. If you have difficulty, contact your instructor asap.
This assignment is in the form of an automatically graded Canvas quiz.